
Restoring the Patient-Clinician Relationship to the Center of Care
The next evolution of patient experience will not be defined by more digital touchpoints. It…

The next evolution of patient experience will not be defined by more digital touchpoints. It will be defined by giving nurses and care teams the capacity to be fully present. The post Restoring the Patient-Clinician Relationship to the Center of Care appeared first on MedCity News.

Prevention requires more than faster detection. Organizations need payment programs with end-to-end visibility and controls that intervene before losses occur. The post From Detection to Defense: Building Fraud-Resilient Healthcare Payment Systems appeared first on MedCity News.

[Sponsored] Healthcare payments are no longer about transactions. It’s about controlling the revenue cycle. And increasingly, that control is not sitting with ISOs. The post The Healthcare Payments Industry Has a Perception Problem appeared first on MedCity News.

Healthcare needs systems that work for providers, for health plans, and most importantly, for patients. And that starts with designing not just for technology, but for the people who depend on it, people who need to trust it will work when it matters most. The post Healthcare IT Isn’t Broken — But How We Design…

[Sponsored] Our new report will focus on how people who work for payers are thinking about AI and cybersecurity needs to protect sensitive healthcare data. The post Take Part in Our Survey on Healthcare AI and Security appeared first on MedCity News.
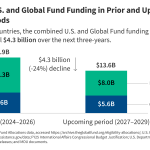
This analysis assesses the magnitude of combined U.S. and Global Fund funding cuts between 2026 and 2029, relative to prior funding levels, in 29 countries with signed MOUs and available data.

The looming impact of federal Medicaid cuts has reignited a long-simmering, costly battle between California’s medical industry and one of its largest health worker unions. SEIU-United Healthcare Workers West, with approximately 120,000 members, has put forward two ballot initiatives to cap the pay of medical executives and require community clinics to spend the vast bulk…

SALT LAKE CITY — Ben Dowse hadn’t expected to treat measles when he became a doctor, but there he was, examining a newborn exposed to the virus in the womb. The infected mother had given birth just hours earlier. The hospital had alerted Dowse to the case before delivery, and he’d braced himself for the…
From the very interesting book Why Fish Don’t Exit by Lulu Miller. To some people, a dandelion might look like a weed, but to others that same plant can be so much more. To an herbalist, it’s a medicine–a way of detoxifying the liver, clearing the skin, and strengthening the eyes. To a painter, it’s…

CMS Administrator Dr. Mehmet Oz outlined the agency’s strategy for making healthcare more affordable during a Tuesday address at the HFMA Annual Conference, touching on everything from Medicare fraud to drug pricing to nutrition. The post The 5 Areas Where Dr. Oz Says CMS Can Make Healthcare More Affordable appeared first on MedCity News.