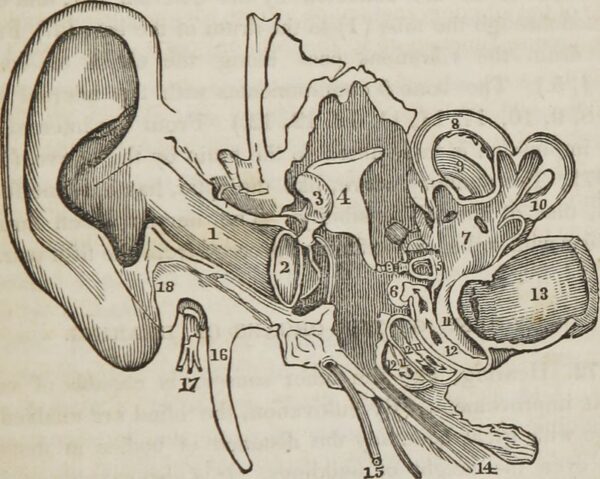
Regeneron Pharmaceuticals’ Otarmeni is now approved for treating hearing loss from an ultra-rare genetic mutation found in an estimated 50 newborns per year. While Regeneron will offer this gene therapy for free, the approval came with a rare pediatric disease priority review voucher that the company can sell for hundreds of millions of dollars.
The post Regeneron Gets Landmark FDA Approval for First Gene Therapy for Hearing Loss appeared first on MedCity News.