
‘2030 Scares Me to Death’: Credit Analysts’ Urgent Warning for Healthcare CFOs
During a panel at HFMA’s annual conference, two healthcare credit analysts said the industry is…

During a panel at HFMA’s annual conference, two healthcare credit analysts said the industry is already fracturing into winners and losers. And with 2030 bringing a perfect storm of demographic pressure, reimbursement cuts and AI disruption, they think window for bold action is closing fast. The post ‘2030 Scares Me to Death’: Credit Analysts’ Urgent…

This budget summary provides an analysis of global health funding amounts provided in the FY 2027 House Labor, Health and Human Services, Education, and Related Agencies (Labor HHS) appropriations bill and accompanying report.

Parabilis Medicines is developing a new type of peptide drug capable of hitting elusive disease targets inside cells. Cancer is the biotech’s initial focus, and the IPO cash will support a lead program zolucatetide, which is in development for a rare type of tumor with limited treatment options. The post Parabilis Medicines’ Quest to Drug…
Officials, environmental health advocates, and skin care industry groups are expressing hope that the Food and Drug Administration’s approval of a sunscreen ingredient on June 9 — after consideration for two decades, and global use for nearly as long — will help restore Americans’ wavering faith in sunscreen. “Bemotrizinol has been used safely in Europe…

The next evolution of patient experience will not be defined by more digital touchpoints. It will be defined by giving nurses and care teams the capacity to be fully present. The post Restoring the Patient-Clinician Relationship to the Center of Care appeared first on MedCity News.

Prevention requires more than faster detection. Organizations need payment programs with end-to-end visibility and controls that intervene before losses occur. The post From Detection to Defense: Building Fraud-Resilient Healthcare Payment Systems appeared first on MedCity News.

[Sponsored] Healthcare payments are no longer about transactions. It’s about controlling the revenue cycle. And increasingly, that control is not sitting with ISOs. The post The Healthcare Payments Industry Has a Perception Problem appeared first on MedCity News.

Healthcare needs systems that work for providers, for health plans, and most importantly, for patients. And that starts with designing not just for technology, but for the people who depend on it, people who need to trust it will work when it matters most. The post Healthcare IT Isn’t Broken — But How We Design…

[Sponsored] Our new report will focus on how people who work for payers are thinking about AI and cybersecurity needs to protect sensitive healthcare data. The post Take Part in Our Survey on Healthcare AI and Security appeared first on MedCity News.
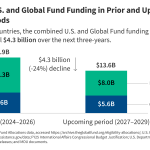
This analysis assesses the magnitude of combined U.S. and Global Fund funding cuts between 2026 and 2029, relative to prior funding levels, in 29 countries with signed MOUs and available data.